【专家述评】|靶向治疗与免疫治疗在放射性碘难治性分化型甲状腺癌中的应用与前景
时间:2025-03-07 12:13:25 热度:37.1℃ 作者:网络
[摘要] 甲状腺癌(thyroid cancer,TC)是一种常见的内分泌系统恶性肿瘤,其中分化型TC(differentiated TC,DTC)占90%以上,通常大多数患者在接受放射性碘(radioactive iodine,RAI)治疗后总体预后良好,但仍有部分患者的病灶在治疗过程中逐渐丧失摄碘能力,成为RAI难治性DTC(radioiodine-refractory DTC,RAIR-DTC),预后较差。对于无法手术切除的RAIR-DTC复发病灶或远处转移灶,既往认为治疗方式有限。随着对RAIR-DTC的发生机制及其生物分子层面的改变有了更深的认识,靶向治疗、免疫治疗及靶向治疗联合免疫治疗呈现出广阔的应用前景,其有效性和安全性也在人类研究中被证实,为RAIR-DTC治疗带来新的希望。本文概括了RAIR-DTC的发生、发展机制、靶向治疗与免疫治疗的临床研究现状及其主要结论,以期为今后的研究提供方向。多激酶抑制剂(multiple kinase inhibitors,MKIs)是晚期转移性RAIR-DTC的一线治疗方案。目前被美国食品药品管理局(Food and Drug Administration, FDA)批准用于治疗RAIR-DTC的药物包括索拉非尼(sorafenib)、仑伐替尼(lenvatinib)、卡博替尼(cabozantinib),前两者也被中国国家药品监督管理局批准用于RAIR-DTC的治疗。中国还有安罗替尼(anlotinib)、多纳非尼(donafenib)被批准用于RAIR-DTC的治疗。以上靶向药物的有效性和安全性均得到了验证。阿帕替尼(apatinib)是一种中国自主研发的血管生成抑制剂,有望成为sorafenib耐药时有效的挽救治疗方法。而单靶点选择性抑制剂因作用靶点单一,不良反应通常较轻。对于某些含有特定突变类型的RAIR-DTC,单靶点选择性抑制剂可以更好地发挥效果。TC通常被认为肿瘤突变负荷(tumor mutation burden,TMB)较低,既往认为免疫治疗的效果有限。包括帕博利珠单抗(pembrolizumab)、度伐利尤单抗(durvalumab)、阿特珠单抗(atezolizumab)、伊匹单抗(ipilimumab)在内的免疫检查点抑制剂,单独使用时疗效常不理想。当联合靶向治疗时,pembrolizumab可以增强靶向药物的疗效,可作为一种可行的挽救疗法,这可能与靶向治疗联合免疫治疗的“协同效应”有关。对于联合治疗,各种治疗分别对抑制肿瘤、延长患者生存期发挥了多大作用需要明确,特别是在病例数较少的情况下,如何设计合理的对照成为关键。既往研究受限于RAIR-DTC定义模糊、纳入患者量少且异质性大,未来需要开展前瞻性、多中心、大规模的临床试验。同时,还需要考虑治疗后延长的无进展生存期(progression-free survival,PFS)是否能转化为长期生存获益,以及是否能改善患者的生活质量。总之,RAIR-DTC的治疗仍面临诸多挑战,未来需要在这些方面不断探索和研究。
[关键词] 放射性碘难治性分化型甲状腺癌;基因突变;信号通路;免疫微环境;靶向治疗;免疫治疗;联合治疗;预后
[Abstract] Thyroid cancer (TC) is a common malignant tumor of the endocrine system, with differentiated TC (DTC) accounting for more than 90%. Most patients usually have a good overall prognosis after receiving radioactive iodine (RAI) treatment, however some patients’ lesions gradually lose the ability to take up iodine during treatment and become RAI-refractory DTC (RAIR-DTC), with a poor prognosis. For RAIR-DTC recurrence lesions or distant metastases that cannot be surgically removed, it was previously believed that there were limited treatment options. With a deeper understanding of the pathogenesis of RAIR-DTC and its changes at the biomolecular level, targeted therapy, immunotherapy and combined targeted and immune therapy have shown broad application prospects. Their effectiveness and safety have also been confirmed in human studies, bringing new hope for the treatment of RAIR-DTC. This article summarized the pathogenesis and development mechanism of RAIR-DTC, the current status of clinical research on targeted therapy and immunotherapy, and their main conclusions, in order to provide direction for future research. Multi-kinase inhibitors (MKIs) are the first-line therapy for advanced metastatic RAIR-DTC. Currently, Food and Drug Administration (FDA) of the United States has approved the following drugs for the treatment of RAIR-DTC: sorafenib, lenvatinib and cabozantinib. The first two of these have been approved by China National Medical Products Administration for RAIR-DTC treatment. In China, anlotinib and donafenib have also been approved for RAIR-DTC treatment. The efficacy and safety of these targeted therapies have been verified. Apatinib, an anti-angiogenesis inhibitor independently developed in China, is expected to be an effective salvage therapy for sorafenib-resistant lesions. Selective single-target inhibitors, with their more specific action targets, generally cause fewer side effects. For certain RAIR-DTC patients with specific mutation types, selective single-target inhibitors may be more effective. TC is generally considered to have a low tumor mutational burden (TMB), and its response to immunotherapy was once thought to be limited. Immune checkpoint inhibitors (ICIs), including pembrolizumab, durvalumab, atezolizumab and ipilimumab, have shown limited efficacy when used alone. However, when combined with targeted therapy, pembrolizumab can enhance the efficacy of targeted drugs, serving as a viable salvage therapy, potentially due to the “synergistic effect” of the combination therapy. It is crucial to determine the individual contributions of each therapy to tumor suppression and survival extension, especially when the sample size is limited. The design of reasonable controls becomes the key in these studies. Previous studies were obstructed by the unclear definition of RAIR-DTC, limited sample sizes and high heterogeneity. Therefore, prospective, multi-center, large-scale clinical trials are needed in the future. Additionally, it is essential to consider whether prolonged progression-free survival (PFS) after treatment can be translated into long-term survival benefits, and whether it improves the quality of life for patients. In conclusion, the treatment of RAIR-DTC still faces many challenges, we must continue to explore and address these issues in the future.
[Key words] Radioiodine-refractory differentiated thyroid cancer; Gene mutation; Signaling pathway; Immune microenvironment; Targeted therapy; Immunotherapy; Combination therapy; Prognosis
1 背景
甲状腺癌(thyroid cancer,TC)是一种常见的内分泌系统恶性肿瘤[1],其中分化型TC (differentiated TC,DTC)占90%以上[2],主要包括甲状腺乳头状癌(papillary thyroid carcinoma,PTC)和甲状腺滤泡癌(follicular thyroid carcinoma,FTC)。另外常见的亚型还有甲状腺髓样癌(medullary thyroid carcinoma,MTC)和甲状腺未分化癌(anaplastic thyroid carcinoma,ATC)。DTC的治疗方法包括手术、促甲状腺激素(thyroid stimulating hormone,TSH)抑制治疗和放射性碘(radioactive iodine,RAI)治疗等。对于无法手术切除及转移性DTC,RAI是首选治疗手段。
尽管大多数患者在接受RAI治疗后总体预后良好,但仍有部分患者的病灶在治疗过程中逐渐丧失摄碘能力,从后续的单一131I治疗中获益的可能性极低。2015年美国甲状腺协会(American Thyroid Association,ATA)指南[3]提出,若DTC符合以下4个特征之一,即可被定义为RAI难治性DTC(RAI-refractory DTC, RAIR-DTC):① 病灶无法摄取RAI;② 病灶早期能摄取RAI,但晚期逐渐失去摄碘能力;③ 病灶为异质性肿瘤(部分细胞能摄取RAI,另一部分不能摄取);④ 病灶能摄取RAI,但疾病仍进展。《放射性碘难治性分化型甲状腺癌诊治管理指南(2024版)》[4]中也对RAIR-DTC进行了定义:① 所有已知DTC病灶均不摄碘;② 尽管部分或全部病灶摄碘,但131I治疗后(1年内)出现疾病进展(progressive disease,PD)。Van Nostrand等[5]提出“碘难治”一词不再意味着病灶永远不会对RAI治疗有反应,因此将患者定义为RAIR-DTC要慎重。目前,马提尼克工作组(Martinique Working Group)正在讨论并重新定义RAIR-DTC的各种分类及关于“碘难治”的术语[5]。
在不额外施加干预的情况下,继续给予RAI治疗对于RAIR-DTC几乎无效。RAIR-DTC患者预后差,总体10年生存率约为10%,中位生存期仅3~5年[6]。因此,探索RAIR-DTC的发病机制,寻找新的治疗方法一直是临床研究的重点。随着分子生物学技术的发展,人们对RAIR-DTC的发生机制及其生物分子层面的改变有了更深的认识。对于无法手术切除的RAIR-DTC复发病灶或远处转移灶,放疗、化疗、再分化治疗、靶向治疗、免疫治疗及放射性粒子植入治疗等逐渐成为临床治疗的新选择。
本综述拟重点总结靶向治疗与免疫治疗在RAIR-DTC中的应用现状、难点及展望。
2 RAIR-DTC的分子生物学及免疫微环境
甲状腺特异性基因是表达甲状腺分化特征的关键基因,这些基因的蛋白产物包括钠碘同向转运体(sodium-iodine symporter,NIS)、甲状腺球蛋白(thyroglobulin,Tg)、促甲状腺激素受体(thyroid stimulating hormone receptor,TSHR)和甲状腺过氧化物酶(thyroid peroxidase,TPO)等。DTC细胞膜上的NIS介导碘摄取,使RAI治疗在DTC细胞中特异性积聚,进而对肿瘤起到杀伤作用[7]。
某些关键基因的突变或重排、某些信号通路异常及表观修饰异常,会使甲状腺特异性基因表达受损、缺失,导致DTC失去甲状腺分化特征,称为“失分化(dedifferentiation)”,其关键环节是NIS表达异常、转运异常或膜定位异常,最终使DTC细胞对RAI的摄取能力减弱,这是RAIR-DTC形成的机制之一。
研究最广泛的通路为丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)通路和磷脂酰肌醇3-激酶(phosphoinositide 3-kinase,PI3K)通路。MAPK通路是调节甲状腺特异性基因的主要通路之一[8],其异常激活能抑制甲状腺特异性基因的表达并使NIS启动子区的组蛋白去乙酰化,最终诱导DTC失分化[9]。BRAF V600E突变是导致MAPK通路异常激活最常见的原因[10],RAS突变[11]和RET重排[12]也是MAPK通路的经典激活剂。PI3K通路异常激活会抑制NIS启动子的转录活性,也是DTC失分化的重要原因[13]。RAS突变[11]和RET重排[14]是PI3K通路的经典激活剂。其他常见的基因突变还包括TRET启动子突变,它与RAIR-DTC的RAI耐药有关,几乎所有伴TERT启动子突变的患者对RAI治疗无效[15]。
免疫微环境与肿瘤发生、发展、侵袭性、耐药性等生物学特点密切相关。DTC的免疫微环境存在多种免疫细胞,包括T细胞、B细胞、肿瘤相关巨噬细胞(tumor-associated macrophage,TAM)、自然杀伤(natural killer,NK)细胞等[16],并且表达程序性死亡蛋白配体-1(programmed death ligand-1, PD-L1)和细胞毒性T淋巴细胞相关抗原4(cytotoxic T lymphocyte antigen 4,CTLA-4)[17]。T细胞表面的程序性死亡蛋白-1(programmed death-1,PD-1)与甲状腺癌细胞表面高表达的PD-L1结合,促进免疫逃逸和增殖[18,19]。PD-L1高表达与DTC患者的淋巴结转移、结外侵袭和低生存率相关[20]。BRAF V600E突变也可导致PD-L1高表达[21]。
基于上述RAIR-DTC的分子生物学及免疫微环境特点,越来越多的靶向治疗和免疫治疗方案正逐步应用于RAIR-DTC,疗效较好且安全性可控。
3 靶向治疗在RAIR-DTC中的应用
3.1 多激酶抑制剂(multiple kinase inhibitors,MKIs)
MKIs是晚期转移性RAIR-DTC的一线治疗方案。目前被美国食品和药物管理局(Food and Drug Administration,FDA)批准用于治疗RAIR-DTC的药物包括索拉非尼(sorafenib)、仑伐替尼(lenvatinib)、卡博替尼(cabozantinib),前两者也被中国国家药品监督管理局批准用于RAIR-DTC的治疗。中国还有安罗替尼(anlotinib)、多纳非尼(donafenib)被批准用于RAIR-DTC的治疗。上述靶向药物相关研究的疗效及常见不良反应详见表1。
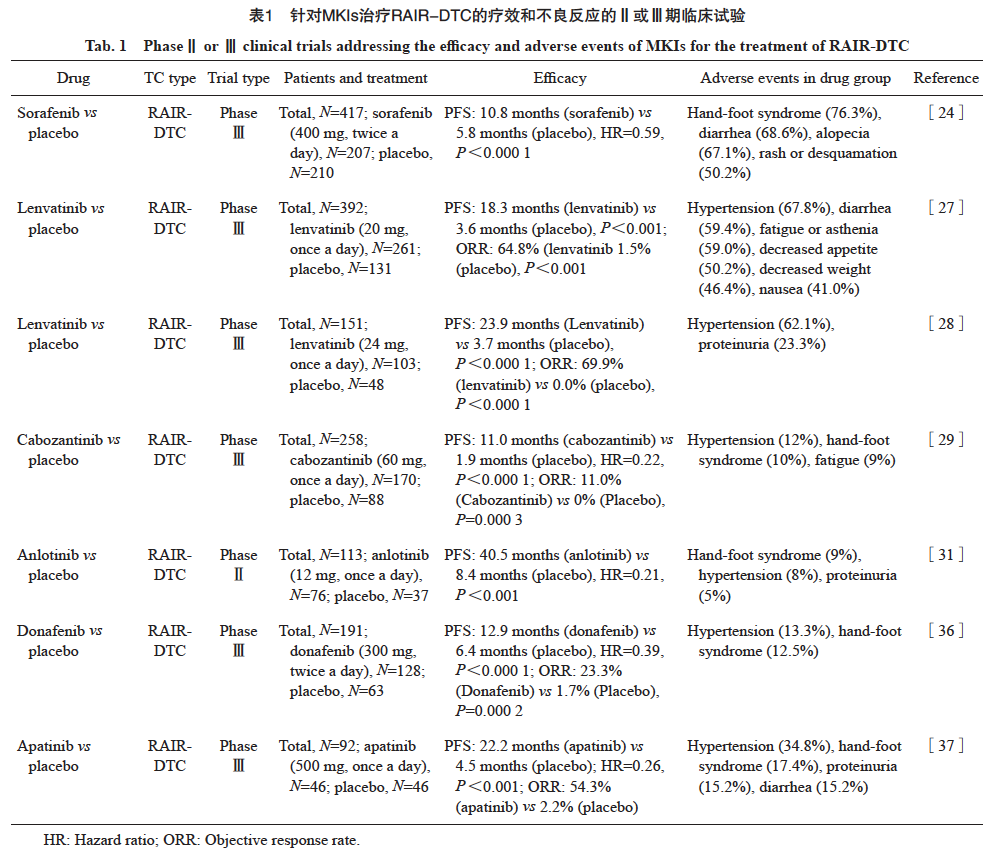
Sorafenib是美国FDA批准用于治疗RAIR-DTC的第1种MKIs,也是目前晚期转移性RAIR-DTC的一线治疗方案,能明显改善预后[22]。Sorafenib的靶点是血管内皮细胞生长因子受体(vascular endothelial growth factor receptor,VEGFR)、RET、RAF和血小板源性生长因子受体β(platelet-derived growth factor receptor β,PDGFRβ)[23]。既往一项大型Ⅲ期临床试验[24]纳入了417例RAIR-DTC患者,发现sorafenib组的中位无进展生存期(progression-free survival, PFS)明显长于安慰剂组。
Lenvatinib[25]和 cabozantinib[26]均为MKIs,被美国FDA批准可作为sorafenib无效时的挽救治疗。Lenvatinib的靶点是VEGFR、成纤维细胞生长因子受体(fibroblast growth factor receptor,FGFR)、PDGFRα、RET、KIT[23]。Cabozantinib的靶点是c-MET、RET、VEGFR[23]。既往一项针对lenvatinib的大型Ⅲ 期临床试验[27]共纳入392例RAIR-DTC患者,lenvatinib组的中位PFS明显长于安慰剂组。中国的一项Ⅲ期临床试验[28]也证明了lenvatinib的有效性和安全性。Lenvatinib因其更长的PFS和更高的客观缓解率(objective response rate,ORR),被美国国立综合癌症网络(National Comprehensive Cancer Network,NCCN)、欧洲肿瘤内科学会(European Society for Medical Oncology,ESMO)、中国临床肿瘤学会(Chinese Society of Clinical Oncology,CSCO)指南优先推荐。既往一项针对cabozantinib的大型Ⅲ期COSMIC-311试验[29]纳入了258例RAIR-DTC患者,cabozantinib组的中位PFS明显长于安慰剂组。2024年一项大型Ⅲ期COSMIC-311试验[30]提示,相当大一部分RAIR-DTC患者会对sorafenib和lenvatinib产生耐药性,但cabozantinib能表现出显著的临床获益,无论患者之前是否接受过sorafenib或lenvatinib治疗。
Anlotinib的靶点是VEGFR、PDGFR、FGFR、c-KIT[23]。中国近期一项大型Ⅱ期临床试验[31]共纳入113例RAIR-DTC患者,anlotinib组的中位PFS明显长于安慰剂组。另外两项回顾性研究[32-33]也认为anlotinib对RAIR-DTC有明确的有效性及良好的安全性,有望在sorafenib治疗失败后发挥重要的补救作用。目前国内有一项Ⅲ 期临床试验(NCT02586337)正在进行,旨在对比anlotinib与安慰剂在RAIR-DTC中的疗效和安全性。
Donafenib可抑制VEGFR等多种受体酪氨酸激酶活性、多种Raf激酶以及下游的Raf/MEK/ERK信号转导通路[34]。一项随机多中心、开放 Ⅱ期临床试验[35]纳入了35例RAIR-DTC成人患者,结果显示,300 mg组在中位PFS和肿瘤缩小趋势方面略优于200 mg组。另一项多中心Ⅲ期临床试验[36]纳入了191例局部晚期/转移性RAIR-DTC患者,结果显示,donafenib较安慰剂显著延长了独立评审委员会评估的中位PFS,在ORR和疾病控制率方面亦均显著优于安慰剂组,且具有良好的耐受性和依从性。Donafenib已获得中国国家药品监督管理局的批准,应用于进展性、局部晚期或转移性RAIR-DTC的治疗。
目前国外有一项关于“ST-1898”(一种新型MKIs)的多中心Ⅱ期临床试验(NCT06359847)正在招募患者,旨在评价ST-1898片剂对一线MKIs治疗后进展的局部晚期或转移性RAIR-DTC的疗效。
3.2 以VEGFR为靶点的抗血管生成药物
阿帕替尼(apatinib)是一种血管生成抑制剂,其靶点是VEGFR-2,但暂未被美国FDA批准用于治疗DTC。林岩松团队的一项Ⅲ期临床试验[37]对比了apatinib与安慰剂在局部晚期或转移性RAIR-DTC患者中的疗效和安全性,共纳入92例RAIR-DTC患者,apatinib组的中位PFS明显长于安慰剂组。该团队的另一项研究[38]发现伴有BRAF V600E突变的进展期RAIR-DTC患者经apatinib治疗后PFS及缓解持续时间较野生组更长,表明BRAF V600E或许可成为潜在的指导酪氨酸激酶抑制剂治疗的生物学标志物。陈立波团队[39]的研究也发现,apatinib是进展期RAIR-DTC的一种新的初始治疗方法,也是当sorafenib耐药时有效的挽救治疗方法。
3.3 单靶点选择性抑制剂
单靶点选择性抑制剂的不良反应较MKIs通常较轻。常见药物包括BRAF抑制剂和MEK抑制剂。
BRAF抑制剂:维莫非尼(vemurafenib)和达拉非尼(dabrafenib)都是单靶点选择性BRAF抑制剂。Dunn等[40]的研究纳入了10例伴BRAF V600E突变的RAIR-DTC患者,给予维莫非尼治疗后4例恢复了RAI摄取,经后续RAI治疗后2例部分缓解(partial response,PR)、2例疾病稳定(stable disease,SD)。Michael Rothenberg等[41]的研究纳入了10例伴BRAF V600E突变的RAIR-DTC患者,给予达拉非尼治疗后6例出现了新的RAI摄取,经后续RAI治疗后2例PR、4例SD。可见BRAF抑制剂需要配合RAI以发挥作用。
MEK抑制剂:司美替尼(selumetinib)是一种高选择性MEK抑制剂,理论上能抑制MEK信号通路从而增加RAI摄取。Ho等[42]的研究纳入了20例RAIR-DTC患者,其中12例出现了RAI摄取增强,超过一半的患者在经后续RAI治疗后疾病PR。
3.4 联合靶向治疗的探索
目前关于双靶联合治疗的研究较少。既往一项Ⅱ期临床试验[43]共纳入了53例BRAF V600E突变阳性的RAIR-DTC患者,26例使用达拉非尼单药治疗、27例使用达拉非尼+曲美替尼联合治疗。但遗憾的是,两组间的疗效并未见显著差异。正在进行的一项Ⅲ期临床试验(NCT04940052)纳入了150例BRAF V600E突变阳性的RAIR-DTC患者,在之前的VEGFR靶向治疗后出现进展。这些患者将以2∶1的比例随机分配到达拉非尼联合曲美替尼组或安慰剂组。期待该试验取得令人满意的结果。另一项正在进行的Ⅰ期临床试验(NCT01947023)旨在探索拉帕替尼(lapatinib)与达拉菲尼联合用药治疗RAIR-DTC的不良反应和最大耐受剂量,并初步记录疗效。
随着对RAIR-DTC分子机制的不断探索,越来越多新型靶向药物或既往曾用于其他恶性肿瘤但其作用尚未在RAIR-DTC中得到验证的靶向药物正逐渐被投入临床试验,靶向治疗的有效性也正逐渐得到临床数据的支持。对于晚期进展期RAIR-DTC无法手术者,靶向治疗是为数不多的选择。
除了考虑疗效,靶向治疗的不良反应也是必须要考虑的问题。针对sorafenib的大型Ⅲ期临床试验[24]中,绝大部分患者出现了不良反应,约66%的患者因不良反应而减少药量、停药甚至退出,sorafenib组有9例患者发生继发性恶性肿瘤(主要为皮肤鳞状细胞癌),另外sorafenib组的死亡人数也明显高于安慰剂组,但不能明确是否为sorafenib直接导致的。上述其他各种靶向药物均有不同程度的不良反应,但总体来说这些不良反应的严重程度和发生比例都在可控的范围内[25,27,29,31,37,39]。
另外,成本效益也是需要考虑的。基于中国的医疗系统,Shi等[44]提出apatinib治疗RAIR-DTC有很好的成本效益,但cabozantinib的成本效益不佳。
因此,对进展期RAIR-DTC的靶向治疗药物的选择需要多方位考量药物的可及性、耐受性、社会经济因素和患者意愿等,从而制订性价比最高、患者获益最大的治疗方案。
4 免疫治疗在RAIR-DTC中的应用
4.1 免疫检查点抑制剂(immune checkpoint inhibitors,ICIs)的应用
ICIs通过阻断免疫检查点分子激活免疫系统攻击肿瘤细胞。ICIs是一类用于肿瘤治疗的免疫药物,其作用机制是通过阻断免疫检查点分子(如PD-1、PD-L1、CTLA-4)等,解除对免疫系统的抑制,从而激活免疫系统,使其能够识别和攻击肿瘤细胞。
甲状腺癌通常被认为是“冷肿瘤”,即肿瘤突变负荷(tumor mutation burden,TMB)较低。高TMB的患者往往对ICIs更敏感,临床疗效也更好[45-46],而甲状腺癌由于TMB低,理论上免疫治疗的效果有限[47]。随着对甲状腺免疫逃避机制的深入研究以及免疫治疗在多种实体恶性肿瘤中的应用突破,甲状腺癌的免疫疗法表现出相当大的治疗潜力。
帕博利珠单抗(pembrolizumab)是目前唯一被批准用于甲状腺癌的ICIs,属于PD-1/PD-L1抑制剂,pembrolizumab通过结合PD-1,阻断PD-1与PD-L1之间的相互作用,从而解除对T细胞功能的抑制,增强T细胞的活性,进而有效地控制和治疗癌症[48]。既往一项pembrolizumab单药的Ⅰ期临床试验[49]纳入了22例PD-L1阳性的RAIR-DTC患者,但仅有2例患者PR。近期一项大型Ⅱ期临床试验[50]纳入了103例之前治疗后进展的DTC患者,随后全部接受pembrolizumab单药治疗,总体ORR仅为6.8%,且与肿瘤PD-L1表达高低无关。最常发生的治疗相关不良反应是疲劳(19.4%)、瘙痒(14.6%)和皮疹(14.6%);13例(12.6%)发生3或4级不良反应,2例(1.9%)因5级不良反应死亡。
度伐利尤单抗(durvalumab)是一种人免疫球蛋白G1κ,通过结合肿瘤细胞上的PD-L1,阻断PD-L1与PD-1及CD80的结合,从而释放T细胞的活性,使得免疫系统能够有效地识别和杀伤肿瘤细胞[51]。一项Ⅰ期临床试验[52]研究了durvalumab与RAI联合治疗复发或转移性TC患者的疗效及安全性,共纳入11例患者,其中2例PR,7例SD,2例PD,且所有患者均未出现严重不良反应,中位PFS为9.8个月。
阿特珠单抗(atezolizumab)是一种人源化单克隆抗体,作为PD-L1阻断剂,其作用机制是能特异地结合肿瘤细胞和肿瘤浸润免疫细胞上的PD-L1,阻断其与T细胞及抗原递呈细胞上的PD-1相互作用,解除PD-1介导的免疫抑制,增强免疫系统对肿瘤细胞的识别和杀伤能力,打破肿瘤细胞的免疫逃逸机制[53]。一项Ⅱ期多中心研究[54]旨在评估atezolizumab用于多种晚期实体瘤(Ⅲ或Ⅳ期)的抗肿瘤活性,其中包括7例PTC和4例FTC患者,尽管该研究提前结束,但结果显示,在18周时,有6例(54.5%)患者癌症未出现进展,ORR达9.1%,已超出试验预设的中期获益标准。
伊匹单抗(ipilimumab)是一种人源化IgG1单克隆抗体,作为一种CTLA-4抑制剂,主要通过与CTLA-4靶向结合,阻断CTLA-4对T细胞的抑制信号,从而使T细胞能够被充分激活,增强免疫系统对肿瘤细胞的杀伤作用[55]。既往一项研究[56]纳入了3例TC(亚型不详)患者,CTLA-4抑制剂单药并未表现出显著的抗肿瘤作用。需要注意的是,该研究并非针对RAIR-DTC。
尽管ICIs单药治疗在特定患者群体中展现出了一定的治疗效果,但其所存在的局限性同样不容忽视。与之形成鲜明对比的是,联合治疗策略不但能够更为有效地激活患者自身的免疫系统,使其产生更为强烈的免疫反应,从而显著增强对肿瘤细胞的杀伤和抑制能力,而且还有望覆盖更为广泛的患者类型,为更多不同疾病特征的患者带来新的治疗契机和希望[57]。一项聚焦于纳武利尤单抗(nivolumab)与ipilimumab联合用药方案用于治疗RAIR-DTC的Ⅱ期临床研究[58]同步开展,在此基础上,还针对ATC和MTC设立了探索性队列研究,该研究纳入了32例DTC、10例ATC和7例MTC患者,DTC、MTC和ATC患者的中位PFS分别为7.11、2.14和4.29个月,2年总生存率分别为77.2%、85.7%和55.6%。结果表明,nivolumab与ipilimumab联用在ATC中具有显著的治疗效果。
4.2 其他免疫疗法
过继性细胞免疫治疗是指从患者体内采集免疫细胞,在体外进行扩增和功能增强,然后将这些经过处理的免疫细胞重新回输到患者体内,以达到直接杀伤肿瘤或激发机体免疫应答的目的。最常用的方法是嵌合抗原受体T(chimeric antigen receptor T,CAR-T)细胞疗法,是通过基因工程技术将CAR引入T细胞表面,使其能够特异性识别肿瘤细胞表面的特定抗原,进而识别并攻击肿瘤细胞[59]。Li等[60]通过细胞和小鼠模型证明了TSHR是CAR-T细胞治疗DTC的潜在靶抗原,抗-TSHR CAR-T细胞治疗可能是癌症局部复发或远处转移患者的治疗选择,但目前未见用于人类DTC的研究报道。一项实验[61]发现CAR-T细胞在体外对ATC细胞有较强的特异性杀伤作用。
过继性细胞免疫治疗还包括肿瘤浸润淋巴细胞(tumor infiltrating lymphocytes,TILs)疗法和NK细胞疗法,但均未见用于人类DTC患者的研究报道。
免疫治疗在RAIR-DTC中的局限性主要表现为单独使用时效果有限,相关研究也较少。未来的方向可能是进一步探索靶向治疗联合免疫治疗,通过不同机制协同作用来提高疗效。同时,还需要解决联合治疗带来的不良反应等问题,并通过前瞻性、多中心、大规模的临床试验来验证其疗效。
5 靶向治疗与免疫治疗联合应用的临床研究现状
以Haugen和French两位学者为主的团队在2020—2024年发表了三项Ⅱ期临床试验的结果:① 2020年5月发表的研究[62]提示与lenvatinib单药相比,lenvatinib联合pembrolizumab对RAIR-DTC患者并未显示出显著优势;② 2020年9月发表的研究[63]显示,24例RAIR-DTC患者在lenvatinib治疗中进展时加用pembrolizumab,17%的患者获得PR;③ 2024年最新发表的研究[64]共纳入57例RAIR-DTC患者,其中30例为初次接受靶向治疗(定义为组1),另外27例为既往接受lenvatinib治疗后进展(定义为组2),入组后均接受lenvatinib(组1剂量固定为20 mg/d;组2剂量为入组前剂量)和pembrolizumab(200 mg/21 d)联合治疗,两组的ORR分别为65.5%和16.0%,提示lenvatinib联合pembrolizumab可能会增强lenvatinib单药治疗的持久性。而对于乐伐替尼治疗后进展的患者,添加pembrolizumab可能是一种可行的挽救疗法。林岩松团队[65]对1例接受一线MKIs治疗后进展的RAIR-DTC患者试验性给予apatinib联合Camrelizumab治疗,呈现出温和且持久的疗效,经过12个月后被评估为PR。
靶向治疗联合免疫治疗从“直接通过相关通路抑制肿瘤生长”和“促进免疫细胞攻击肿瘤细胞”两种不同的机制来抑制肿瘤,理论上疗效会优于单药靶向治疗或单药免疫治疗。更有研究[66]认为靶向治疗和免疫治疗之间存在“协同效应”,靶向药物诱导肿瘤细胞死亡裂解,导致新抗原的释放,可能会提高ICIs的疗效。既往有研究[67]将BRAF抑制剂维莫非尼与CTLA-4抑制剂用于治疗转移性黑色素瘤,提出维莫非尼可能通过增加细胞毒性T细胞的识别[68]和降低肿瘤免疫抑制[69]的方式提高CTLA-4抑制剂的疗效。靶向治疗联合免疫治疗在RAIR-DTC中也有广阔的应用前景。
实际上,靶向治疗联合免疫治疗的效果并非一定优于单药,现有的研究主要受限于纳入患者数量过少,今后需要前瞻性、多中心、大规模的临床试验来验证。另外,患者是否能同时承受两种治疗带来的不良反应也是需要考虑的问题。
6 总结与展望
目前,对于RAIR-DTC的治疗,一线治疗方案如sorafenib、lenvatinib等MKIs以及新获批的donafenib已被证实能明显改善患者预后,二线治疗方案如cabozantinib、anlotinib等在一线方案疗效不佳时也取得了理想效果,且不良反应在可控范围内。
既往曾认为,理论上甲状腺癌对免疫治疗的反应有限,既往大型临床试验也认为ICIs单药对RAIR-DTC疗效不佳,但靶向治疗联合免疫治疗相较于靶向单药却可能发挥出明显更优的疗效,可能与靶向治疗联合免疫治疗的“协同效应”有关。对于联合治疗(无论是双靶联合还是靶向治疗联合免疫治疗,或其他联合治疗),各种治疗分别对抑制肿瘤、延长患者生存期发挥了多大作用需要明确,特别是在病例数较少的情况下,如何设计合理的对照成为关键。
未来需要解决的问题包括选择合适的治疗药物、确定最佳剂量、明确何时开始及结束治疗、有效管理不良反应等。此外,目前针对RAIR-DTC没有明确的定义和诊断标准,患者间异质性大,纳入和排除标准的制定需要非常慎重。大部分研究纳入的患者量都较少,因此需要开展前瞻性、多中心、大规模的临床试验。同时,还需要考虑治疗后延长的PFS是否能转化为长期生存获益,以及是否能改善患者的生活质量。另外,成本效益、患者的治疗意愿及依从性等也应纳入考虑范围。总之,RAIR-DTC的治疗仍面临诸多挑战,未来的研究需要在这些方面不断探索。
第一作者:
林秋玉,博士,副主任医师、副教授。
通信作者:
林承赫,博士,主任医师、教授,吉林大学第一医院核医学科主任。
作者贡献声明:
林秋玉:资料收集,文章撰写和修改;王宇鑫:资料收集,文章撰写;林承赫:主题指导,文章撰写和修改。
[参考文献]
[1] SIEGEL R L, MILLER K D, FUCHS H E, et al. Cancer statistics, 2022[J]. CA Cancer J Clin, 2022, 72(1): 7-33.
[2] MAO Y S, XING M Z. Recent incidences and differential trends of thyroid cancer in the USA[J]. Endocr Relat Cancer, 2016, 23(4): 313-322.
[3] HAUGEN B R, ALEXANDER E K, BIBLE K C, et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer[J]. Thyroid, 2016, 26(1): 1-133.
[4] 中国临床肿瘤学会核医学专家委员会, 中国临床肿瘤学会甲状腺癌专家委员会, 中华医学会核医学分会, 等. 放射性碘难治性分化型甲状腺癌诊治管理指南(2024版)[J].中华核医学与分子影像杂志, 2024, 44(6): 359-372.
Nuclear Medicine Expert Committee of Chinese Society of Clinical Oncology, Thyroid Cancer Expert Committee of Chinese Society of Clinical Oncology, Chinese Society of Nuclear Medicine, et al. Management guidelines for radioactive iodinerefractory differentiated thyroid cancer (2024 edition)[J]. Chin J Nucl Med Mol Imag, 2024, 44(6): 359-372.
[5] VAN NOSTRAND D, VEYTSMAN I, KULKARNI K, et al. Redifferentiation of differentiated thyroid cancer: clinical insights from a narrative review of literature[J]. Thyroid, 2023, 33(6): 674-681.
[6] BROSE M S, SMIT J, CAPDEVILA J, et al. Regional approaches to the management of patients with advanced, radioactive iodine-refractory differentiated thyroid carcinoma[J]. Expert Rev Anticancer Ther, 2012, 12(9): 1137-1147.
[7] YAVUZ S, PUCKETT Y. Iodine-131 uptake study[M], StatPearls. Treasure Island: StatPearls Publishing, 2023.
[8] HOU P, BOJDANI E, XING M Z. Induction of thyroid gene expression and radioiodine uptake in thyroid cancer cells by targeting major signaling pathways[J]. J Clin Endocrinol Metab, 2010, 95(2): 820-828.
[9] ZHANG Z J, LIU D X, MURUGAN A K, et al. Histone deacetylation of NIS promoter underlies BRAF V600Epromoted NIS silencing in thyroid cancer[J]. Endocr Relat Cancer, 2014, 21(2): 161-173.
[10] NAGARAJAH J, LE M N, KNAUF J A, et al. Sustained ERK inhibition maximizes responses of Braf V600E thyroid cancers to radioiodine[J]. J Clin Invest, 2016, 126(11): 4119-4124.
[11] LAHA D, NILUBOL N, BOUFRAQECH M. New therapies for advanced thyroid cancer[J]. Front Endocrinol (Lausanne), 2020, 11: 82.
[12] HAROON AL RASHEED M R, XU B. Molecular alterations in thyroid carcinoma[J]. Surg Pathol Clin, 2019, 12(4): 921-930.
[13] XING M Z. Identifying genetic alterations in poorly differentiated thyroid cancer: a rewarding pursuit[J]. J Clin Endocrinol Metab, 2009, 94(12): 4661-4664.
[14] MIZUKAMI T, SHIRAISHI K, SHIMADA Y, et al. Molecular mechanisms underlying oncogenic RET fusion in lung adenocarcinoma[J]. J Thorac Oncol, 2014, 9(5): 622-630.
[15] YANG X, LI J, LI X Y, et al. TERT promoter mutation predicts radioiodine-refractory character in distant metastatic differentiated thyroid cancer[J]. J Nucl Med, 2017, 58(2): 258-265.
[16] ARAQUE K A, GUBBI S, KLUBO-GWIEZDZINSKA J. Updates on the management of thyroid cancer[J]. Horm Metab Res, 2020, 52(8): 562-577.
[17] TUCCILLI C, BALDINI E, SORRENTI S, et al. CTLA-4 and PD-1 ligand gene expression in epithelial thyroid cancers[J]. Int J Endocrinol, 2018, 2018: 1742951.
[18] CUNHA L L, MARCELLO M A, VASSALLO J, et al. Differentiated thyroid carcinomas and their B7H1 shield[J]. Future Oncol, 2013, 9(10): 1417-1419.
[19] AL-ABDALLAH A, JAHANBANI I, MEHDAWI H, et al. Down-regulation of the human major histocompatibility complex class Ⅰ chain-related gene A (MICA) and its receptor is mediated by microRNA-146b-5p and is a potential mechanism of immunoediting in papillary thyroid carcinoma[J]. Exp Mol Pathol, 2020, 113: 104379.
[20] ULISSE S, TUCCILLI C, SORRENTI S, et al. PD-1 ligand expression in epithelial thyroid cancers: potential clinical implications[J]. Int J Mol Sci, 2019, 20(6): 1405.
[21] BASTOS A U, OLER G, NOZIMA B H N, et al. BRAF V600E and decreased NIS and TPO expression are associated with aggressiveness of a subgroup of papillary thyroid microcarcinoma[J]. Eur J Endocrinol, 2015, 173(4): 525-540.
[22] CORRADO A, FERRARI S M, POLITTI U, et al. Aggressive thyroid cancer: targeted therapy with sorafenib[J]. Minerva Endocrinol, 2017, 42(1): 64-76.
[23] SHEN H Z, ZHU R, LIU Y Y, et al. Radioiodine-refractory differentiated thyroid cancer: molecular mechanisms and therapeutic strategies for radioiodine resistance[J]. Drug Resist Updat, 2024, 72: 101013.
[24] BROSE M S, NUTTING C M, JARZAB B, et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: a randomised, double-blind, phase 3 trial[J]. Lancet, 2014, 384(9940): 319-328.
[25] DACOSTA BYFIELD S A, ADEJORO O, COPHER R, et al. Real-world treatment patterns among patients initiating small molecule kinase inhibitor therapies for thyroid cancer in the United States[J]. Adv Ther, 2019, 36(4): 896-915.
[26] SILAGHI H, LOZOVANU V, GEORGESCU C E, et al. State of the art in the current management and future directions of targeted therapy for differentiated thyroid cancer[J]. Int J Mol Sci, 2022, 23(7): 3470.
[27] SCHLUMBERGER M, TAHARA M, WIRTH L J, et al. Lenvatinib versus placebo in radioiodine-refractory thyroid cancer[J]. N Engl J Med, 2015, 372(7): 621-630.
[28] ZHENG X Q, XU Z G, JI Q H, et al. A randomized, phase Ⅲ study of lenvatinib in Chinese patients with radioiodinerefractory differentiated thyroid cancer[J]. Clin Cancer Res, 2021, 27(20): 5502-5509.
[29] BROSE M S, ROBINSON B G, SHERMAN S I, et al. Cabozantinib for previously treated radioiodine-refractory differentiated thyroid cancer: Updated results from the phase 3 COSMIC-311 trial[J]. Cancer, 2022, 128(24): 4203-4212.
[30] CAPDEVILA J, KRAJEWSKA J, HERNANDO J, et al. Increased progression-free survival with cabozantinib versus placebo in patients with radioiodine-refractory differentiated thyroid cancer irrespective of prior vascular endothelial growth factor receptor-targeted therapy and tumor histology: a subgroup analysis of the COSMIC-311 study[J]. Thyroid, 2024, 34(3): 347-359.
[31] CHI Y, ZHENG X Q, ZHANG Y, et al. Anlotinib in locally advanced or metastatic radioiodine-refractory differentiated thyroid carcinoma: a randomized, double-blind, multicenter phase Ⅱ trial[J]. Clin Cancer Res, 2023, 29(20): 4047-4056.
[32] 李 娇, 韩 娜, 卢承慧, 等. 安罗替尼治疗远处转移性放射性碘难治性分化型甲状腺癌的有效性及安全性分析[J].中华核医学与分子影像杂志, 2023, 43(8): 470-474.
LI J, HAN N, LU C H, et al. Efficacy and safety analysis of anlotinib in the treatment of distant metastatic radioactive iodine-refractory differentiated thyroid cancer[J]. Chin J Nucl Med Mol Imag, 2023, 43(8): 470-474.
[33] 张芳蕾, 翟红彦, 闫瑞红, 等. 安罗替尼对进展期放射性碘难治性分化型甲状腺癌的疗效及对病灶摄碘功能的影响[J]. 中华核医学与分子影像杂志, 2024, 44(10): 592-596.
ZHANG F L, ZHAI H Y, YAN R H, et al. Efficacy of anlotinib in the treatment of advanced radioactive iodine-refractory differentiated thyroid cancer and the effect on iodine uptake of lesions[J]. Chin J Nucl Med Mol Imag, 2024, 44(10): 592-596.
[34] LIU Y J, WANG J F, HU X P, et al. Radioiodine therapy in advanced differentiated thyroid cancer: resistance and overcoming strategy[J]. Drug Resist Updat, 2023, 68: 100939.
[35] LIN Y S, YANG H, DING Y, et al. Donafenib in progressive locally advanced or metastatic radioactive iodine-refractory differentiated thyroid cancer: results of a randomized, multicenter phase II trial[J]. Thyroid, 2021, 31(4): 607-615.
[36] LIN Y S, QIN S K, YANG H, et al. Multicenter randomized double-blind phase Ⅲ trial of donafenib in progressive radioactive iodine-refractory differentiated thyroid cancer[J]. Clin Cancer Res, 2023, 29(15): 2791-2799.
[37] LIN Y S, QIN S K, LI Z Y, et al. Apatinib vs placebo in patients with locally advanced or metastatic, radioactive iodine-refractory differentiated thyroid cancer: the REALITY randomized clinical trial[J]. JAMA Oncol, 2022, 8(2): 242-250.
[38] 刘杰蕊, 张 鑫, 孙郁青, 等. BRAF V600E突变辅助预测放射性碘难治性分化型甲状腺癌阿帕替尼治疗效果的意义[J]. 中华核医学与分子影像杂志, 2023, 43(8): 465-469.
LIU J R, ZHANG X, SUN Y Q, et al. Significance of BRAF V600E mutation in prediction of the efficacy of apatinib for radioactive iodine-refractory differentiated thyroid cancer\n[J]. Chin J Nucl Med Mol Imag, 2023, 43(8): 465-469.
[39] QIU X, CHENG L, SA R, et al. Initial or salvage treatment with apatinib shows promise against radioiodine-refractory differentiated thyroid carcinoma[J]. Eur Thyroid J, 2022, 11(2): e210065.
[40] DUNN L A, SHERMAN E J, BAXI S S, et al. Vemurafenib redifferentiation of BRAF mutant, RAI-refractory thyroid cancers[J]. J Clin Endocrinol Metab, 2019, 104(5): 1417-1428.
[41] MICHAEL ROTHENBERG S, MCFADDEN D G, PALMER E L, et al. Redifferentiation of iodine-refractory BRAF V600Emutant metastatic papillary thyroid cancer with dabrafenib[J]. Clin Cancer Res, 2015, 21(5): 1028-1035.
[42] HO A L, GREWAL R K, LEBOEUF R, et al. Selumetinibenhanced radioiodine uptake in advanced thyroid cancer[J]. N Engl J Med, 2013, 368(7): 623-632.
[43] BUSAIDY N L, KONDA B, WEI L, et al. Dabrafenib versus dabrafenib+trametinib in BRAF-mutated radioactive iodine refractory differentiated thyroid cancer: results of a randomized, phase 2, open-label multicenter trial[J]. Thyroid, 2022,
32(10): 1184-1192.
[44] SHI B, MA W B, PAN H S, et al. Cost-effectiveness of apatinib and cabozantinib for the treatment of radioiodine-refractory differentiated thyroid cancer[J]. Front Pharmacol, 2022, 13: 860615.
[45] WANG X S, LI M Y. Correlate tumor mutation burden with immune signatures in human cancers[J]. BMC Immunol, 2019, 20(1): 4.
[46] PINHEIRO NETO A, LUCCHESI H L, VALSECCHI V A D S, et al. Immunotherapy for patients with thyroid cancer: a comprehensive appraisal[J]. Chin Clin Oncol, 2024, 13(3):
36.
[47] NIKIFOROV Y E, NIKIFOROVA M N. Molecular genetics and diagnosis of thyroid cancer[J]. Nat Rev Endocrinol, 2011, 7(10): 569-580.
[48] WANG S M, KHAN F I. Investigation of molecular interactions mechanism of pembrolizumab and PD-1[J]. Int J Mol Sci, 2023, 24(13): 10684.
[49] MEHNERT J M, VARGA A, BROSE M S, et al. Safety and antitumor activity of the anti-PD-1 antibody pembrolizumab in patients with advanced, PD-L1-positive papillary or follicular thyroid cancer[J]. BMC Cancer, 2019, 19(1): 196.
[50] OH D Y, ALGAZI A, CAPDEVILA J, et al. Efficacy and safety of pembrolizumab monotherapy in patients with advanced thyroid cancer in the phase 2 KEYNOTE-158 study[J]. Cancer, 2023, 129(8): 1195-1204.
[51] PAZ-ARES L, DVORKIN M, CHEN Y B, et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial[J]. Lancet, 2019, 394(10212): 1929-1939.
[52] BURMAN B, SHERMAN E J, KRIPLANI A, et al. Radioiodine (RAI) in combination with durvalumab for recurrent/metastatic thyroid cancers[J]. J Clin Oncol, 2020, 38(15_suppl): 6587.
[53] WÄCHTER S, KNAUFF F, ROTH S, et al. Synergic induction of autophagic cell death in anaplastic thyroid carcinoma[J]. Cancer Invest, 2023, 41(4): 405-421.
[54] TABERNERO J, ANDRE F, BLAY J Y, et al. Phase Ⅱ multicohort study of atezolizumab monotherapy in multiple advanced solid cancers[J]. ESMO Open, 2022, 7(2): 100419.
[55] WANG Y C, ZHANG H, LIU C, et al. Immune checkpoint modulators in cancer immunotherapy: recent advances and emerging concepts[J]. J Hematol Oncol, 2022, 15(1): 111.
[56] SAKAMURI D, GLITZA I C, BETANCOURT CUELLAR S L, et al. Phase I dose-escalation study of anti-CTLA-4 antibody ipilimumab and lenalidomide in patients with advanced cancers[J]. Mol Cancer Ther, 2018, 17(3): 671-676.
[57] BARBARI C, FONTAINE T, PARAJULI P, et al. Immunotherapies and combination strategies for immunooncology[J]. Int J Mol Sci, 2020, 21(14): 5009.
[58] LORCH J H, BARLETTA J A, NEHS M, et al. A phase II study of nivolumab (N) plus ipilimumab (I) in radioidine refractory differentiated thyroid cancer (RAIR DTC) with exploratory cohorts in anaplastic (ATC) and medullary thyroid cancer (MTC)[J]. J Clin Oncol, 2020, 38(15_suppl): 6513.
[59] SHENG X Y. Evaluation of safety and therapeutic efficacy of CAR-T therapy[J]. Highlights Sci Eng Technol, 2024, 91: 272-276.
[60] LI H N, ZHOU X, WANG G, et al. CAR-T cells targeting TSHR demonstrate safety and potent preclinical activity against differentiated thyroid cancer[J]. J Clin Endocrinol Metab, 2022, 107(4): 1110-1126.
[61] MIN I M, SHEVLIN E, VEDVYAS Y, et al. CAR T therapy targeting ICAM-1 eliminates advanced human thyroid tumors[J]. Clin Cancer Res, 2017, 23(24): 7569-7583.
[62] HAUGEN B, FRENCH J, WORDEN F P, et al. Lenvatinib plus pembrolizumab combination therapy in patients with radioiodine-refractory (RAIR), progressive differentiated thyroid cancer (DTC): results of a multicenter phase Ⅱ International Thyroid Oncology Group trial[J]. J Clin Oncol, 2020, 38(15_suppl): 6512.
[63] HAUGEN B, FRENCH J D, WORDEN F, et al. Pembrolizumab salvage add-on therapy in patients with radioiodine-refractory (RAIR), progressive differentiated thyroid cancer (DTC) progressing on lenvatinib: results of a multicenter phase Ⅱ International Thyroid Oncology Group trial[J]. Ann Oncol, 2020, 31: S1086-S1087.
[64] FRENCH J D, HAUGEN B R, WORDEN F P, et al. Combination targeted therapy with pembrolizumab and lenvatinib in progressive, radioiodine-refractory differentiated thyroid cancers[J]. Clin Cancer Res, 2024, 30(17): 3757-3767.
[65] LI J Y, ZHANG X, MU Z Z, et al. Response to apatinib and camrelizumab combined treatment in a radioiodine refractory differentiated thyroid cancer patient resistant to prior antiangiogenic therapy: a case report and literature review[J]. Front Immunol, 2022, 13: 943916.
[66] VANNEMAN M, DRANOFF G. Combining immunotherapy and targeted therapies in cancer treatment[J]. Nat Rev Cancer, 2012, 12(4): 237-251.
[67] ACKERMAN A, KLEIN O, MCDERMOTT D F, et al. Outcomes of patients with metastatic melanoma treated with immunotherapy prior to or after BRAF inhibitors[J]. Cancer, 2014, 120(11): 1695-1701.
[68] BONI A, COGDILL A P, DANG P, et al. Selective BRAFV600E inhibition enhances T-cell recognition of melanoma without affecting lymphocyte function[J]. Cancer Res, 2010, 70(13): 5213-5219.
[69] SUMIMOTO H, IMABAYASHI F, IWATA T, et al. The BRAFMAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells[J]. J Exp Med, 2006, 203(7): 1651-1656.


